Patient recruitment strategy is often treated as a single decision: which vendor to hire. In practice, it's a set of choices made across the enrollment lifecycle, each of which affects how many patients enter the trial, how quickly they move through the pipeline, and how many remain through completion.
Below are 15 strategies that appear across well-run recruitment programs, with context on when each tends to be most useful.
1. Map your two patient populations before the trial opens.
Every trial draws from two sources: patients already in the site's panel and patients who need to be identified through outreach. The first group enrolls quickly but depletes. The second requires more infrastructure but sustains performance longer. Knowing the approximate size of each population by site informs your recruitment plan and budget before the trial opens.
2. Start recruitment planning at protocol development, not after enrollment stalls.
The administrative overhead of engaging external recruitment support — vendor selection, contracting, IRB amendments, site training, system integration — typically requires 8 to 12 weeks. Starting the process 3 to 4 months before First Patient First Visit means your infrastructure is operational when enrollment begins. Starting after enrollment has plateaued means you've already lost months.
3. Treat the phone screen as a conversion funnel, not an administrative step.
The first phone interaction between a site and a prospective patient may be the highest-stakes 5 minutes of your entire recruitment timeline. Sites that conduct two or three calls before scheduling a screen visit tend to convert qualified patients at lower rates. Patient interest drops substantially after 72 hours without follow-up, and continues to decline each subsequent day.
Standardizing the phone screen across sites, training CRCs on patient-centered communication, and setting a follow-up timing standard are among the more straightforward operational improvements available.
4. Use digital channels for patient identification, not just awareness.
Social media advertising, targeted digital campaigns, and patient community platforms can identify eligible patients who would not be reached through physician referral or site outreach alone. In one example from our network, a single week of social media advertising generated seven qualified patient inquiries for a trial that had been behind on enrollment.
The channel is mature enough that "should we use digital?" is generally not the right question. The more relevant questions are which platforms, which targeting approach, and which messaging resonates in your therapeutic area.
5. Build rapid follow-up into your site workflows.
Research consistently suggests that patient interest in trial participation drops substantially within 24 to 72 hours of initial contact if no follow-up occurs. This isn't primarily a motivation problem. It's a logistical one. Patients who express interest are managing competing demands, and momentum dissipates quickly.
Establishing a standard that inbound inquiries receive a follow-up contact within 24 hours, and training site staff accordingly, tends to meaningfully improve conversion rates from inquiry to screen visit.
6. Screen patients against multiple protocols when possible.
Inclusion and exclusion criteria are often more limiting in practice than they appear on paper. For a patient with a given condition, the probability of being eligible for any single trial may be relatively low. Screening against multiple relevant protocols at a single encounter substantially increases the probability that the patient can be placed somewhere.
For sponsors running multiple studies in an indication, this has direct operational implications. For recruitment platforms, it's a core value proposition.
7. Segment sites by performance and concentrate resources accordingly.
Site performance varies substantially even within a single trial. Some sites enroll consistently above projection. Others struggle to enroll at all. Tufts CSDD data suggests roughly 48% of activated sites fall short of their enrollment targets.
Rather than distributing referral volume and support evenly, identifying high-performing sites early and concentrating resources there generally produces better overall enrollment than trying to rescue every underperforming site simultaneously.
8. Monitor pipeline depth, not just enrollment numbers.
Enrollment numbers reflect what happened 60 days ago. The metric that predicts future performance is the number of patients currently moving through each stage of the screening process — what can be thought of as Patients in Process. A thin pipeline today means a shortfall in enrollment 8 weeks from now.
Building visibility into pipeline depth at the site level gives teams lead time to respond before the gap appears in enrollment data.
9. Move from batch reporting to real-time site performance monitoring.
Weekly or monthly site performance reports are often too lagged to enable meaningful operational response. By the time a sponsor identifies an underperforming site through standard reporting, weeks of enrollment window have passed.
Real-time visibility into screening rates, conversion rates, and referral volume by site enables dynamic reallocation of resources, faster identification of site-level issues (staffing changes, screening backlogs, protocol interpretation questions), and more precise enrollment forecasting.
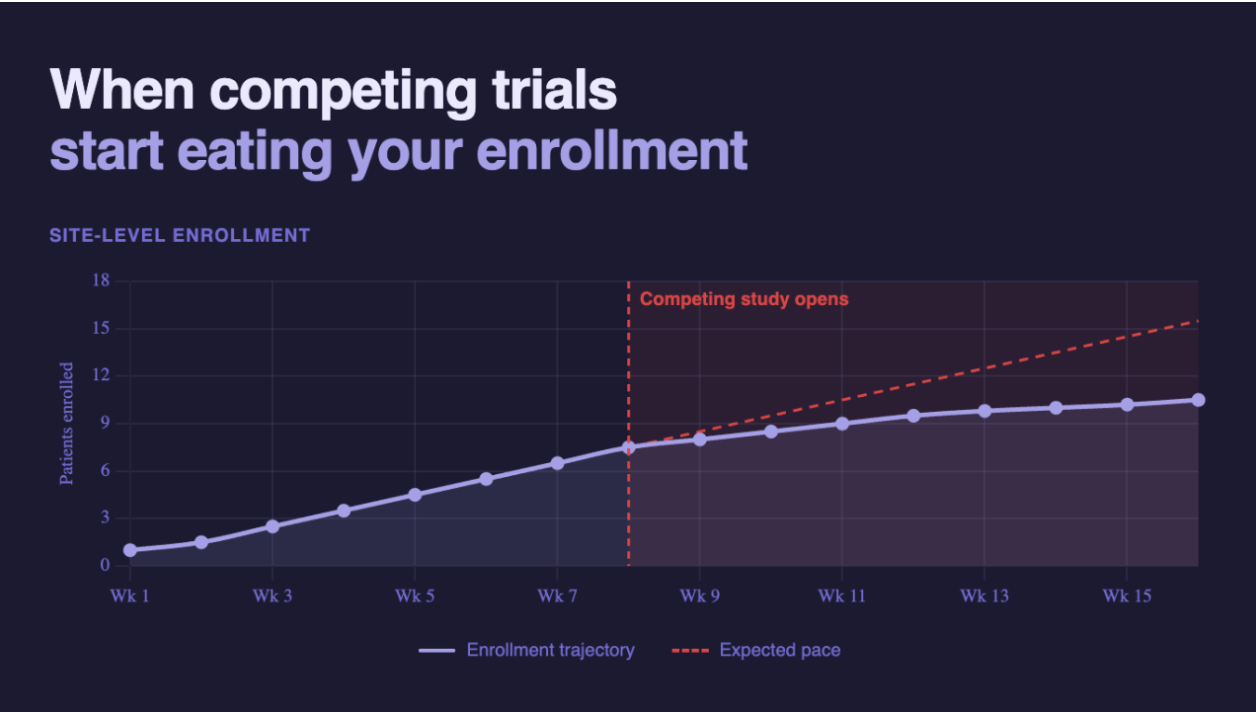
10. Plan for the month 3–6 enrollment plateau explicitly.
The enrollment plateau that appears around month 3 to 6 in many trials is not random. It reflects the depletion of the established patient panel, the operational learning curve at new sites, and the lag inherent in the recruitment pipeline.
Treating it as a predictable structural feature — and planning external patient identification infrastructure to activate before it occurs — is more effective than treating it as a performance failure to be corrected reactively.
11. Use EMR-based patient matching to expand the eligible pool.
Electronic medical record matching platforms can surface patients within a health system who meet protocol criteria but haven't been identified by the clinical team. This shifts the identification step earlier and tends to produce leads with higher baseline eligibility rates than broad advertising channels.
The operational consideration is that EMR-based matching requires IRB approval and data use agreements that take time to establish — reinforcing the value of early planning.
12. Reduce patient burden in protocol design when possible.
One of the most common reasons sites give for enrollment challenges: patients can't meet the visit frequency the protocol requires. Once or twice weekly visits represent a substantial burden for most working adults. Where the science permits, reducing visit frequency, enabling remote assessments, or offering travel reimbursement can meaningfully expand the reachable patient population.
These are design-level decisions, which is why recruitment considerations benefit from being represented in protocol development rather than only at the operations stage.
13. Build patient-facing communication into your site workflows.
A significant source of dropout occurs not during screening but during the study itself, when patients feel disconnected from the research process. CRCs managing heavy administrative loads often cannot maintain consistent patient communication.
Building structured touchpoints into the protocol — including visit reminders, progress updates, and clear advance notice of what each visit involves — tends to support retention without requiring substantial additional staff time.
14. Model the financial downside of recruitment failure before the trial opens.
Teams that haven't explicitly modeled the cost of timeline extension often underinvest in recruitment infrastructure. A 4-month enrollment delay in a mid-stage neuroscience program may represent $6M or more in direct operational costs and over $24M in NPV impact from delayed market entry.
Against those numbers, recruitment support that prevents meaningful delay tends to be a favorable investment — one that is easier to justify prospectively than retroactively.
15. Evaluate recruitment partners on speed-to-first-enrollment, not just stated capability.
Most recruitment vendors can describe their platform capabilities. The more operationally relevant question is: how long from IRB approval to your first enrolled patient?
First enrollment within 6 to 8 weeks of IRB approval is achievable with the right infrastructure in place. Vendors who cannot give you a concrete answer to this question — or whose historical data suggests longer timelines — should be evaluated accordingly, particularly for programs with aggressive enrollment targets.
Power is a centralized patient recruitment platform for neuropsychiatry and adjacent therapeutic area trials. Learn more at withpower.com.