The AtaiBeckley merger reflects a bet that shorter treatment durations will improve patient access and payer acceptance.
AtaiBeckley emerged on November 4, 2025 from the combination of atai Life Sciences and Beckley Psytech, united by a conviction that shorter treatment durations will reshape how psychedelic therapies reach patients.
The merger, announced in June 2025, was explicitly contingent on Phase 2b results. When that data came through in July showing meaningful efficacy and durability, it cleared the path for the companies to complete their strategic combination.
In separate conversations on our podcast (Power to the Patients), both Srinivas Rao (now CEO of AtaiBeckley) and Cosmo Feilding Mellen (CEO of Beckley Psytech, now on the Board of Directors at AtaiBeckley) articulated strikingly similar strategic reasoning despite coming from vastly different backgrounds. Their alignment on the commercial and clinical logic of shorter-duration treatments illuminates why this merger made strategic sense beyond the Phase 2b data.
The Phase 2b Data That Supported the Hypothesis
The strategic combination was explicitly contingent on positive Phase 2b results. When those results came through in July, showing that single-dose BPL-003 produced statistically significant and durable MADRS reductions versus control in treatment-resistant depression patients, it cleared the path for the companies to officially combine.
BPL-003, an intranasal formulation of 5-MeO-DMT (mebufotenin benzoate), demonstrated statistically significant and durable antidepressant effects in treatment-resistant depression patients. At Day 29, the 8mg dose showed a mean MADRS reduction from baseline of 12.1 points compared to 5.8 points in the 0.3mg control group (p=0.0025) — a 6.3-point advantage. The 12mg dose showed an 11.1-point baseline reduction (p=0.0038) — a 5.3-point advantage over control. Both doses produced statistically significant improvements as early as Day 2, with effects maintained through Week 8.
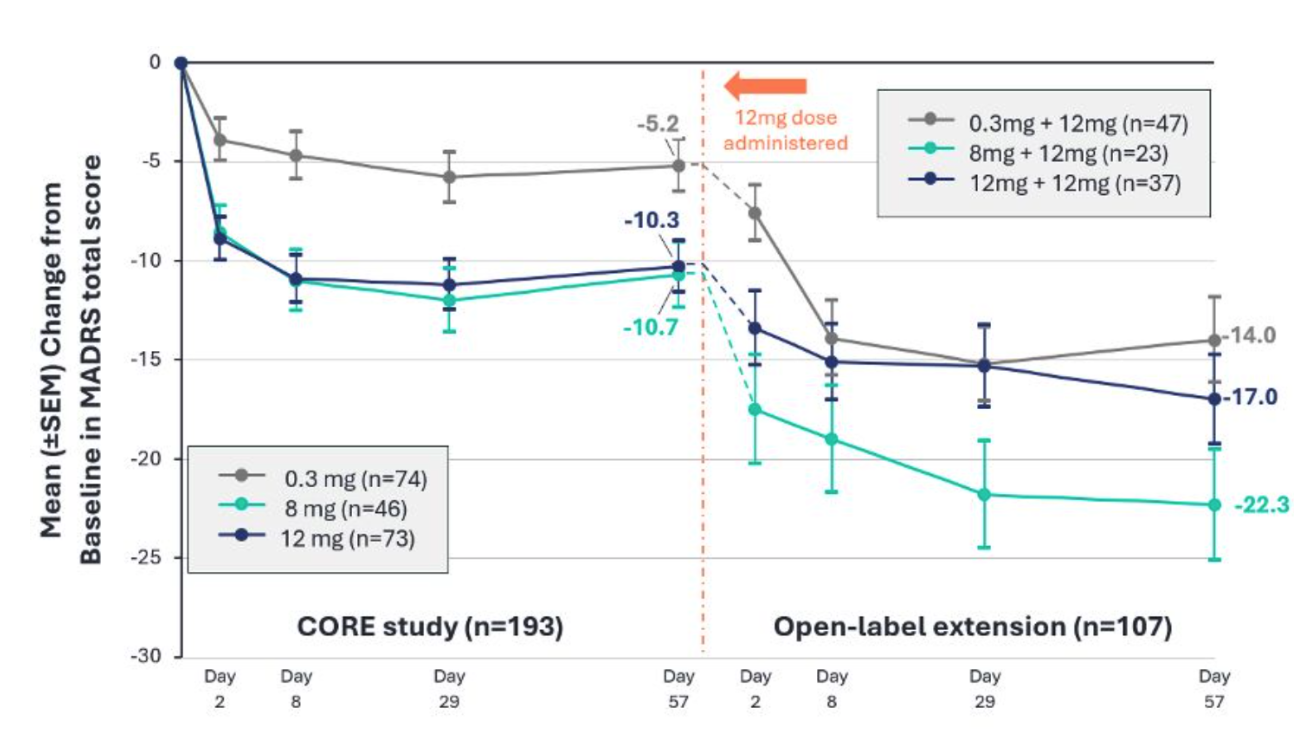
Critically for patient access: the majority of participants met discharge readiness criteria at the 90-minute assessment, with average time to meet discharge readiness criteria within two hours across all treatment arms.
The data met the merger's pre-specified success criteria, earned FDA Breakthrough Therapy designation, and secured an End-of-Phase 2 meeting. Phase 3 initiation is planned for Q2 2026.
But the clinical results tell only part of the story. The strategic rationale, rooted in making treatment actually work for patients' lives and clinicians' practices, reveals why duration became the defining feature of the bet both companies made.
Engineering Meets Heritage
"I'm brutally pragmatic," Srinivas Rao, now CEO of AtaiBeckley, explained on our podcast. "The thing that they've heard repeatedly is KISS… keep it simple, stupid."
Rao's engineering background shaped atai's approach from the beginning. When evaluating DMT formulation options, the question wasn't just pharmacological — it was operational. "How do you get something the patients are going to deal with well, the doctors are going to deal with well, that is easy to manufacture?" he said. The team considered needles and other administration methods before settling on the oral thin film (for VLS-01, their DMT buccal program) and intranasal spray (for BPL-003).
Cosmo Feilding Mellen, CEO of Beckley Psytech and now on the Board at AtaiBeckley, brought a different lens — one shaped by 25 years of Beckley Foundation research. "I was arguably born into it," he noted, describing how his mother's pioneering work in psychedelic science created a foundation of credibility and early-stage research that most startups couldn't replicate.
"What we were trying to think is how could we improve upon that offering and make it more accessible to patients," Feilding Mellen explained when describing Beckley's decision to pursue 5-MeO-DMT. The logic was straightforward: "If we can show that we can get similar levels of safety and efficacy that the first generation drugs like oral psilocybin are getting but the treatment takes an hour or two rather than a whole day, that becomes a much more scalable and accessible treatment."
Why Shorter Works: The Patient and Provider Case
Both CEOs converged on the same insight: treatment duration isn't just an operational detail — it helps determine whether patients will actually access treatment and whether clinicians will offer it.
"For a clinician, do you want to be monitoring a patient for one to two hours or do you want to be monitoring a patient for eight hours?" Feilding Mellen asked. The answer shapes so much, from staffing requirements to how many patients a clinic can realistically treat per day.
For patients, the visit reduction is material. Spravato's protocol requires twice-weekly sessions for a month, then weekly or biweekly maintenance. If BPL-003's durability holds in Phase 3, patients might require just a handful of annual visits. That's the difference between treatment fitting into a normal life versus organizing life around treatment.
For working patients, 50+ annual visits means negotiating time off, arranging childcare, or managing complex logistics that make treatment inaccessible regardless of insurance coverage. Four visits transforms the access equation entirely.
Rao framed it through infrastructure economics. Traditional antidepressants require reaching 25,000-30,000 prescribing physicians — a 2,500-3,000 person sales force. Spravato's specialized clinic model changed that calculus. "There's 4,500 clinics so far," Rao noted in his podcast interview. Current figures show the network has grown to over 5,000 REMS-certified sites. "Even if that doubles, this is a much smaller kind of sales force." He estimates J&J uses no more than 100-200 reps for Spravato.
This isn't theoretical positioning — AtaiBeckley is building to fit existing infrastructure. "We could more or less get dragged and dropped into the infrastructure that they've created," Rao said, referencing J&J's two-hour REMS window that BPL-003 fits within. Rather than asking patients and providers to adapt to psychedelic therapy, they're adapting psychedelic therapy to how clinics already operate.
Starting With the Hardest Patients First
The decision to pursue treatment-resistant depression rather than broader major depressive disorder reflects another strategic calculation. "The more resistant patients are those that are most at risk, they're also the ones that cost the most to the healthcare system," Feilding Mellen explained. "Therefore, regulators are more likely to accept going into that more difficult patient population and insurers and state health providers are more likely to pay for those treatments."
It's a risk-benefit and cost-benefit calculation: tackle the hardest-to-treat patients where unmet need is highest, then expand the label if successful.
The market validation for this approach already exists. J&J's Spravato, targeting the same treatment-resistant depression population, generated $459 million in Q3 2025 sales — an annualized run rate of $1.8 billion. The company projects peak sales of $3-3.5 billion annually by 2027-2028. This commercial success came despite modest efficacy (approximately 4-point MADRS improvement versus placebo) and potentially requiring over 30-50 visits annually depending on individual dosing schedules.
If Spravato can achieve blockbuster status with those characteristics, a treatment showing 5-6 point MADRS improvements and requiring approximately four annual visits presents a fundamentally different value proposition to patients, providers, and payers.
The Path Forward: From Phase 3 to Patients
AtaiBeckley has positioned itself at a critical inflection point. With cash runway extending into 2029 — through the anticipated first Phase 3 readout for BPL-003 — the company has the resources to execute on its central bet: that shorter-duration treatments can succeed where longer protocols have struggled to scale.
"We are bringing together the assets, expertise and vision of atai Life Sciences and Beckley Psytech to transform patient outcomes," Rao said in the merger announcement. "Around the world, too many people continue to suffer without effective treatments and AtaiBeckley is taking a decisive step toward changing this."
The merged company has made a calculated decision to concentrate resources on three Phase 2 programs rather than expanding the portfolio. "You've got a finite amount of resources, there's only so many people in the company," Rao acknowledged. Focus over proliferation.
The End-of-Phase 2 meeting with FDA in the coming months will determine Phase 3 trial design for BPL-003. If all proceeds as planned, the first Phase 3 trial will launch in Q2 2026, with the combined entity's fate hinging on whether 90-minute treatments can demonstrate comparable efficacy to eight-hour protocols in pivotal trials.
But the strategic vision extends beyond proving clinical efficacy. Both CEOs, speaking independently on our podcast months before the merger closed, articulated the same insight: the challenge isn't just developing effective psychedelics — it's developing psychedelics that patients will actually access, clinicians will actually adopt, and healthcare systems will actually reimburse.
"I want to figure out what is absolutely the cleanest and fastest path," Rao said, a philosophy that now defines the combined company's approach.
For Feilding Mellen, the question was also about real-world implementation: "If we can show that we can get similar levels of safety and efficacy that the first generation drugs like oral psilocybin are getting but the treatment takes an hour or two rather than a whole day, that becomes a much more scalable and accessible treatment."
The psychedelic drug development sector has spent years proving these compounds work. AtaiBeckley is betting the next decade will be won by whoever proves they work in the real world — where patients have jobs and families, clinicians manage full schedules, and payers scrutinize cost-effectiveness.
The merger united an engineer's "keep it simple" philosophy with 25 years of research pedigree. Phase 3 will test whether that combination can bring psychedelic medicine from clinical trials to clinical practice.
Now comes the pivotal test: can shorter work at scale?
Brandon Li · Co-Founder, Power
For clinical operations teams working in neuropsychiatry: Talk to our team about what we're learning from sponsors at the frontier.



